Tag: bioequivalence
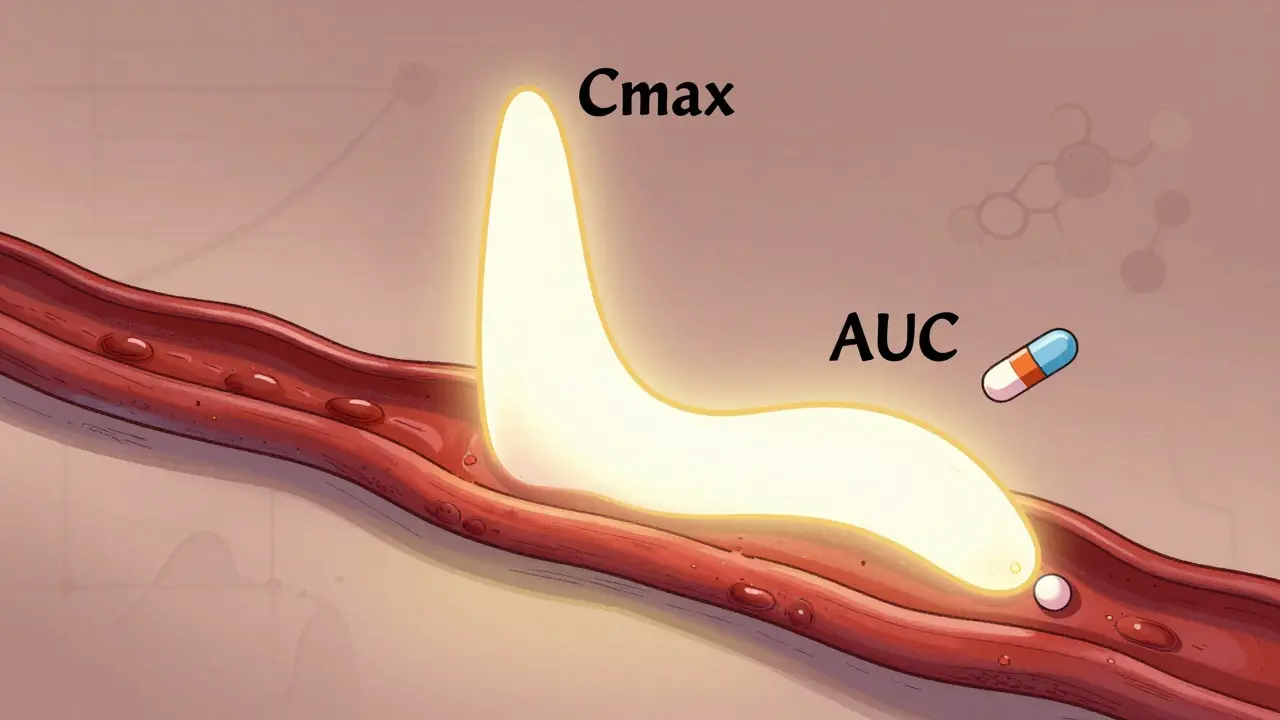
Cmax and AUC in Bioequivalence: Peak Concentration and Systemic Exposure Explained
Cmax and AUC are the two key pharmacokinetic measures used globally to prove generic drugs work like brand-name versions. Cmax shows peak concentration, AUC shows total exposure-both must meet strict 80%-125% criteria for approval.

Reassurance from Research: What Clinical Studies Say About Switching from Brand to Generic Medications
Clinical studies show that while most generic medications work as well as brand-name drugs, some-especially for epilepsy and heart conditions-can cause problems after switching. Learn what the data says and how to protect your health.

Medical Education on Generics: Do Doctors Learn Equivalence?
Doctors prescribe generics 90% of the time, but many still doubt their effectiveness. Why? Medical education rarely teaches bioequivalence. This is how we fix it.