When a generic drug hits the market, you might wonder: is it really the same as the brand-name version? The answer lies in two numbers: Cmax and AUC. These aren’t just lab jargon-they’re the backbone of how regulators decide if a generic drug works just as well as the original. If you’ve ever been told your prescription was switched to a cheaper version, this is why that switch is safe.
What Cmax Tells You About Drug Absorption
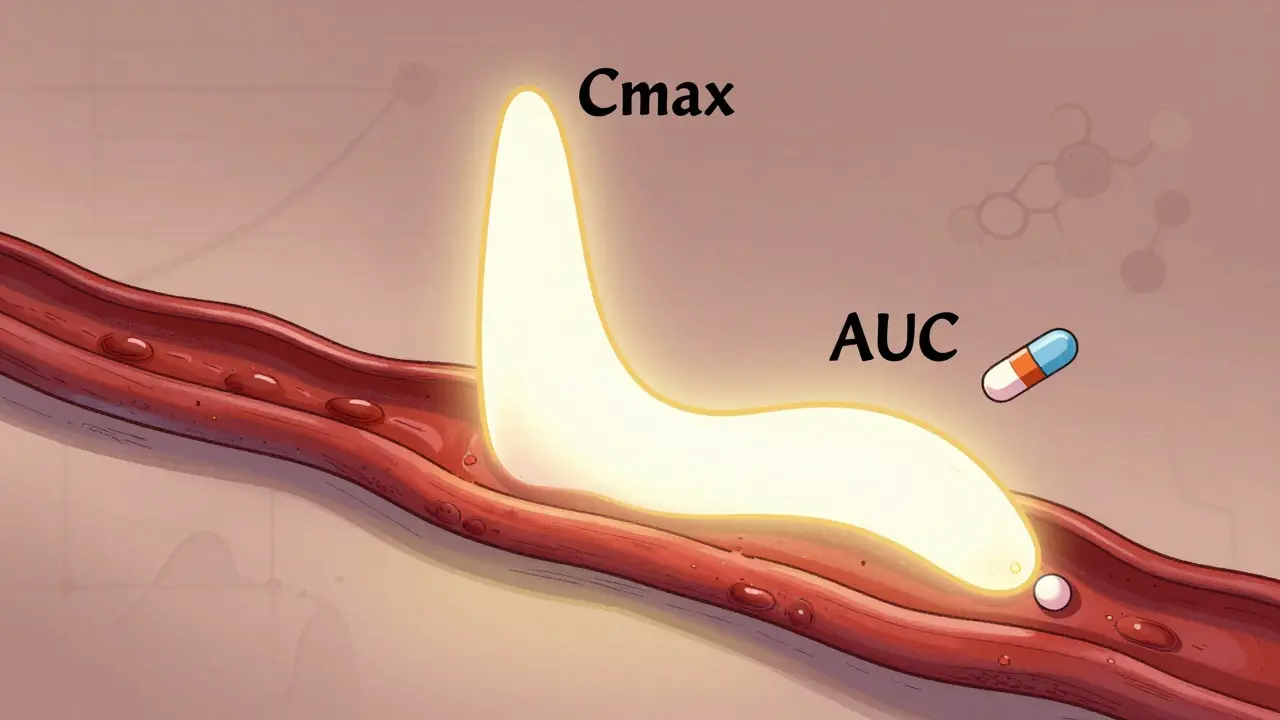
Cmax stands for maximum plasma concentration. Think of it as the highest point a drug reaches in your bloodstream after you take it. It’s measured in milligrams per liter (mg/L) and tells you how fast the drug gets absorbed. For some medicines, this speed matters a lot. Take painkillers like ibuprofen: if the peak is too low or too slow, you won’t feel relief. For drugs like digoxin or warfarin, a Cmax that’s too high could mean dangerous side effects.
Regulators don’t just look at the number-they look at timing too. Tmax, or time to reach Cmax, helps figure out how quickly the drug enters your system. A generic drug might have a slightly different Tmax than the original, but as long as Cmax falls within acceptable limits, it’s still considered equivalent. Studies show that for most immediate-release tablets, Cmax should be within 80% to 125% of the brand-name version. That means if the original hits 10 mg/L, the generic can range from 8 to 12.5 mg/L and still be approved.
Why this range? It’s not arbitrary. It’s based on decades of clinical data showing that differences smaller than 20% rarely affect how well a drug works or how safe it is. The key is consistency. If a generic consistently hits within this range across hundreds of volunteers, regulators approve it.
What AUC Reveals About Total Drug Exposure
AUC, or area under the curve, measures the total amount of drug your body is exposed to over time. It’s calculated by plotting blood concentration levels from the moment you take the drug until it’s almost completely gone. The unit? Milligram-hours per liter (mg·h/L). This number tells you how much drug your body absorbs overall-not just how high it peaks, but how long it stays there.
For drugs that need to work continuously-like antibiotics, antihypertensives, or seizure medications-AUC is often more important than Cmax. If your body doesn’t get enough total exposure, the drug won’t be effective. AUC is especially critical for drugs with a narrow therapeutic index, where even a small drop in exposure can lead to treatment failure. For example, if a patient’s AUC for levothyroxine drops by 15%, they might develop hypothyroidism without realizing why.
Unlike Cmax, which is a single point, AUC is a cumulative measure. It’s shaped by how well the drug dissolves, how fast it passes through the gut, and how quickly the liver breaks it down. Two drugs might have the same Cmax but very different AUCs. That’s why both numbers are required. One tells you about speed; the other tells you about total dose.
Why Both Cmax and AUC Are Required
You might think: if AUC shows total exposure, why do we even need Cmax? Because drugs don’t just need to be absorbed-they need to be absorbed in the right way.
Imagine two versions of the same drug. One releases slowly over 12 hours. The other bursts all at once. They might have the same AUC-same total exposure-but very different Cmax values. The fast-release version could cause nausea or dizziness because of a sudden spike. The slow one might be too weak to work at all if the peak is too low.
Regulatory agencies like the FDA and EMA don’t just look at one metric-they require both. The rules are clear: both Cmax and AUC must fall within the 80%-125% range. If one passes and the other fails, the generic is rejected. This isn’t bureaucracy-it’s safety. A 2019 analysis of 42 studies in JAMA Internal Medicine found no meaningful difference in outcomes between generics and brand-name drugs only when both parameters met the standard.
There are exceptions. For drugs with high variability-where the same person’s response changes a lot from day to day-regulators sometimes use tighter limits or adjusted methods. But even then, Cmax and AUC remain the starting point. The system is built on them because they’ve been tested, validated, and proven over 40 years.
How Bioequivalence Studies Work
Before a generic drug is approved, it goes through a bioequivalence study. These aren’t long-term clinical trials-they’re tightly controlled, short-term tests usually done in 24 to 36 healthy volunteers.
Here’s how it works: participants take the brand-name drug in one period, then the generic in another, with a washout period in between. Blood is drawn 12 to 18 times over 24 to 72 hours, depending on the drug’s half-life. The samples are analyzed using ultra-sensitive tools like LC-MS/MS, which can detect drug levels as low as 0.1 nanograms per milliliter.
Why so many samples? Because missing even one key time point can ruin the Cmax estimate. If the first blood draw after dosing is at 30 minutes, but the drug peaks at 45 minutes, the Cmax could be underestimated. That’s why guidelines stress using actual sampling times-not scheduled ones. Industry data shows that about 15% of failed bioequivalence studies are due to poor sampling design.
The data is then log-transformed (because drug concentrations follow a log-normal distribution), and statistical models calculate the 90% confidence interval for the ratio of geometric means. If both AUC and Cmax ratios fall between 0.8 and 1.25, the drugs are declared bioequivalent.
What Happens When the Numbers Don’t Match
Not every generic passes. Sometimes, differences in formulation-like the type of filler, coating, or manufacturing process-change how the drug dissolves. A tablet that breaks down too slowly won’t reach the right Cmax. A capsule that dissolves too fast might spike the concentration too high.
When a study fails, the manufacturer has to go back to the drawing board. They might reformulate the drug, change the manufacturing process, or run another study. In 2022, over 1,200 generic applications were approved in the U.S. alone, but many more were rejected or delayed. That’s why not every generic looks identical-even if it’s the same active ingredient.
There’s also a myth that generics are “weaker.” That’s not true. When they meet Cmax and AUC standards, they deliver the same amount of drug, at the same rate, as the brand. The difference isn’t in potency-it’s in how the drug is packaged. The inactive ingredients might vary, but those don’t affect how the active drug works.

The Bigger Picture: Why This Matters
Bioequivalence testing saves lives and money. Without Cmax and AUC, every generic drug would need expensive clinical trials to prove it works. Instead, we rely on pharmacokinetics-measurable, repeatable science. This system lets patients access life-saving drugs at a fraction of the cost. In 2022, generics accounted for over 90% of all prescriptions in the U.S., saving billions.
And it’s not just the U.S. The same standards are used in the EU, Canada, Australia, Japan, and over 120 countries. The World Health Organization recognizes Cmax and AUC as the global gold standard. Even with new technologies like modeling and simulation on the horizon, these two metrics remain the foundation. As one FDA scientist put it: “We’ve had 30 years to test this. It works.”
What’s Changing in the Future
Regulators are starting to look beyond Cmax and AUC for complex drugs-like extended-release formulations or those with multiple absorption peaks. The FDA’s 2023 draft guidance suggests using partial AUC (e.g., exposure in the first 2 hours) for these cases. For narrow therapeutic index drugs like warfarin, some agencies now recommend tighter limits: 90% to 111% instead of 80% to 125%.
But even with these updates, Cmax and AUC aren’t going away. They’re too well understood, too validated, and too critical. For the vast majority of drugs-oral tablets, capsules, syrups-these two numbers are still the most reliable way to ensure patients get the right medicine, every time.
Why do regulators require both Cmax and AUC instead of just one?
Cmax tells you how fast a drug reaches its highest level in the blood, which affects how quickly it starts working and whether it causes side effects. AUC tells you how much of the drug your body absorbs over time, which affects how well it works overall. A drug could have the same total exposure (AUC) as the original but peak too high or too low (Cmax), leading to safety or effectiveness issues. That’s why both are required.
Can a generic drug have a different Tmax and still be bioequivalent?
Yes. Tmax (time to reach peak concentration) is considered supportive, not decisive. As long as Cmax and AUC fall within the 80%-125% range, a small difference in Tmax is acceptable. For example, if the brand peaks at 2 hours and the generic at 1.5 hours, that’s usually fine-unless it’s a drug where timing is critical, like some seizure medications.
Is the 80%-125% range the same everywhere in the world?
Most major regulators-including the FDA, EMA, and Health Canada-use the same 80%-125% range. Some countries, like those following WHO guidelines, adopt it too. However, for drugs with high variability or narrow therapeutic windows, some agencies allow tighter limits (e.g., 90%-111%) after additional review. The standard remains consistent across most markets.
Do Cmax and AUC apply to all types of drugs?
They apply to most oral drugs, especially immediate-release formulations. For injectables, inhalers, or topical products, different methods are used. Modified-release drugs with complex absorption patterns may need additional metrics, like partial AUC. But for the vast majority of pills and capsules you take, Cmax and AUC are the gold standard.
Why is logarithmic transformation used in bioequivalence calculations?
Drug concentrations in the blood don’t follow a normal distribution-they follow a log-normal pattern. That means the data is skewed, and using regular averages would give misleading results. Taking the natural logarithm of Cmax and AUC values makes the data symmetric and allows accurate statistical comparison. This is required by both FDA and EMA guidelines.
Understanding Cmax and AUC doesn’t just help you understand generic drugs-it helps you trust them. These numbers aren’t hidden behind lab doors; they’re the reason you can confidently take a cheaper pill and know it will work just as well.







Ray Foret Jr.
March 8, 2026 AT 15:35Samantha Fierro
March 10, 2026 AT 12:22Jazminn Jones
March 11, 2026 AT 06:42Stephen Rudd
March 13, 2026 AT 04:41APRIL HARRINGTON
March 15, 2026 AT 00:30Leon Hallal
March 15, 2026 AT 19:31Judith Manzano
March 17, 2026 AT 04:03rafeq khlo
March 19, 2026 AT 02:51Morgan Dodgen
March 20, 2026 AT 00:34Philip Mattawashish
March 20, 2026 AT 19:31Tom Sanders
March 22, 2026 AT 04:47George Vou
March 22, 2026 AT 17:18Katy Shamitz
March 24, 2026 AT 03:22Nicholas Gama
March 24, 2026 AT 14:18Mary Beth Brook
March 26, 2026 AT 03:18